 This review highlights the impact that iPSC-based cell therapies can have, should development thereof gain more traction. Disorders of the circulatory, as well as nervous and visual systems, were all studied in equal measure. Conversely, Japan was the frontrunner in terms of conducting therapeutic trials, and the average number of participants was much lower, at roughly 29. In terms of nontherapeutic trials, the USA conducted the majority, a large average number of participants-187-was included in the trials, and studies on circulatory system disorders comprised a slightly higher proportion of total studies. Our main observations included global distribution, purpose, target size, and types of disorder relevant to evaluated trials. Comparisons were firstly drawn between observational and interventional studies before the latter was further analyzed in terms of therapeutic and nontherapeutic trials. Data were derived both from well-known registries recording clinical trials from across the globe, and databases from individual countries. Herein, our aim was to determine trends in globally conducted clinical trials involving iPSCs. However, despite the immense cell therapy potential, relatively few clinical trials evaluating iPSC-technology have actually translated into interventional, clinically applied treatment regimens. 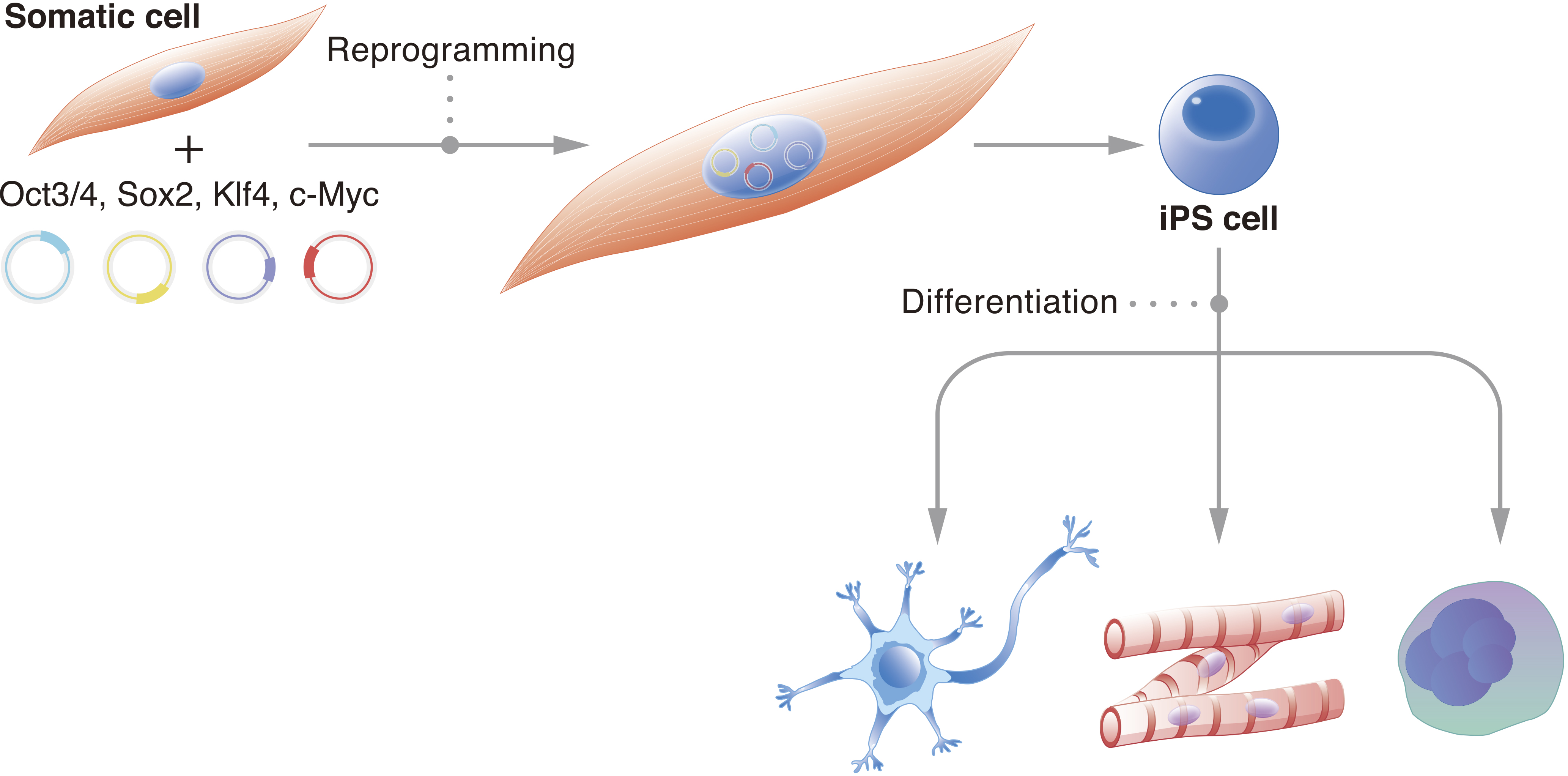
In 2006, the induced pluripotent stem cell (iPSC) was presented to the world, paving the way for the development of a magnitude of novel therapeutic alternatives, addressing a diverse range of diseases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
